“Super bugs” and “food safety.” You can say the two in one breath simply because they are so closely connected.
“Super bugs” is a popularized term for “antibiotic resistance,” or “antimicrobial resistance.” They don’t go by that name for nothing. According to the federal Centers for Disease Control and Prevention, each year in the United States, at least 2.8 million people are infected with antibiotic-resistant bacteria or fungi. More than 35,000 people die as a result. Antibiotic resistant bacteria frequently show up in outbreak strains of pathogens such as E. Coli, Salmonella and Listeria monocytogenes.
When looking ahead, the picture only gets more daunting. Some medical experts predict that worldwide by 2050 the number of deaths attributed to antibiotic resistance could reach 10 million and cost trillions of dollars — unless collective action is taken on a global scale.
No wonder then that some human and animal health experts are referring to this as a “slow moving pandemic.”
What in the world is this all about?
Antibiotics, often referred to as “miracle drugs,” are medicines that kill or stop the growth of bacteria, which is a good thing. That applies to humans and animals alike. But in the case of livestock and poultry, especially at large-scale operations such as industrial farms, antibiotics are often overused in ways that promote drug resistance to them. This can happen when low doses are routinely added to feed and water to promote growth and to prevent health problems caused by overcrowding, and unsanitary conditions. That’s different from using them to treat a specific medical problem.
In this conflict between bacteria and antibiotics, resistance happens when the bacteria develop the ability to evade the antibiotics designed to kill them. That means the germs continue to grow and to outcompete other drugs — potentially leading to infections that are harder to treat. Thus the name “super bugs.”
To see an animation about how bacteria become resistant to antibiotics, click here.
As a result of the over use of antibiotics, some dangerous strains of foodborne bacteria found in animals and poultry, such as E. coli, Salmonella and S. aureus are now resistant to multiple antibiotics. If a person becomes infected with an antibiotic-resistant strain of a foodborne pathogen, what was once, in many cases, an easy-to-treat disease can quickly develop into a life-threatening illness — one that requires expensive medical care and can even lead to death.
But what does this have to do with food safety?
According to the U.S. Center for Disease Control and Prevention, when animals are slaughtered and processed for food, antibiotic-resistant bacteria, if present in the animal, can contaminate meat or other animal products. Microscopic amounts of animal waste can also carry antibiotic-resistant bacteria.
Fruits and vegetables can become contaminated in many ways, including through contact with soil or water containing waste from animals.
People can get foodborne infections in different ways:
- From handling or eating meat, seafood, milk or eggs that are raw or undercooked and contaminated with resistant bacteria;
- From handling or eating fruits and vegetables contaminated with resistant bacteria;
- From contact with animal waste, either directly or when it gets into water and the environment;
- From touching or caring for animals without proper handwashing;
- From breathing in dust particles downwind of livestock farms or feedlots on which drug-resistant bacteria are hitching a ride.
In the case of food safety, foodborne antibiotic-resistant bacteria have already made their way into the food chain, which can make curing people sickened with the bacteria all that more challenging.

According to a 2013 CDC report, of the 18 drug-resistant organisms highlighted as “alarming,” four are potentially fatal foodborne bacteria: Campylobacter, E. coli, Salmonella and Shigella.
The report also points out that resistant bacteria in food-producing animals are of particular concern because these animals serve as carriers. Resistant bacteria can contaminate the foods that come from those animals, and people who consume these foods can develop antibiotic-resistant infections.
Bottomline, animals — like humans — carry bacteria, including antibiotic resistant bacteria, in their gut. When meat animals and poultry are slaughtered and processed, these bacteria can contaminate the meat or other products. In addition, the people handling these products can then be infected. And so can the people and produce crops downwind or downstream from where these animals are raised.
Over time, warns the Food and Drug Administration’s (FDA) Center for Veterinary Medicine, the use of antibiotics and other antimicrobial drugs will further complicate health care professionals’ efforts to select the appropriate antibiotic for treatment.
This issue of antibiotic resistance applies to humans and animals alike, especially considering that humans rely on some of the same antibiotics — referred to as “medically important” antibiotics — as animals to cure health problems. Some of these problems in humans are pneumonia, meningitis, and strep throat.
As in the case of livestock health, these medically important antibiotics cannot be misused or overused without running the risk of having a patient develop antibiotic resistant pathogens.
Examples of medically important antibiotics are penicillins, tetracyclines, erythromycins, cephalosporins and fluoroquinolones.
Although the CDC points out that the majority of drug-resistant infections occur in hospitals, it also says that there’s growing concern over antibiotic-resistant infections from food.
Also, according to the CDC, 75 percent of dangerous new infections, including pandemics, spill over from animals to human populations. These animal-borne threats include viruses as well as new forms of antibiotic resistance genes and the multi-drug resistant superbugs that carry them.
Pointing out that effective antibiotics have been one of the pillars allowing people to live longer, be healthier and benefit from modern medicine, Dr. Keiji Fukuda, formerly a Special Representative for Antimicrobial Resistance for the Director-General at the World Health Organization (WHO), put it this way: “Without urgent coordinated action by many stakeholders, the world is headed for a post-antibiotic era, in which common infections and minor injuries that have been treatable for decades can once again kill.”
“Unless we take significant actions to improve efforts to prevent infections and also change how we produce, prescribe and use antibiotics, the world will lose more and more of these global public health goods, and the implications will be devastating,” he warns.
Tackling an out-of-kilter problem
For decades now, U.S. policymakers, as well as medical and veterinary professionals, have sent out warnings about the rising threat from superbugs.
All in all, the general public believes that “surely something is being done about this,” especially since it’s such a serious health issue.
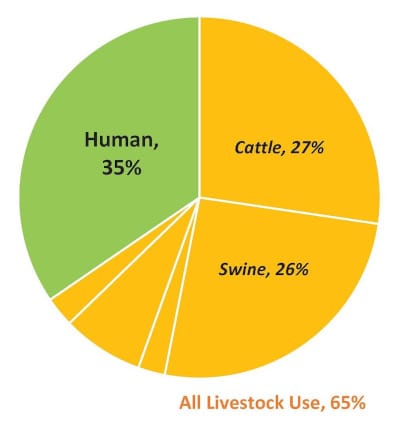
But consider this: According to recent research done by the Center For Disease Dynamics, Economics & Policy and the National Resources Defense Council (NRDC), the use of antibiotics in livestock is rising while medical use in humans is falling.
“Our analysis shows 44 percent more medically important drugs are destined for cattle and swine than for human medical use,” says a website developed by the two associations.
In coming up with this conclusion, they joined forces to fill in what they see as a “critical data gap.” They did this by presenting a side-by-side comparison of antibiotic use in animals and humans in the United States over time. In a post, they supply charts and graphs highlighting their findings.
“In the United States, we don’t track antibiotic use very well,” said Dr. David Wallinga, senior health officer at NRDC and one of the authors of the blog, in an interview with Food Safety News. “FDA does report each year on antibiotics sold for use in livestock, including those that are medically important. In terms of the latter, our latest blog showed that nearly twice as many antibiotics are being sold for use in poultry and livestock as are sold for human medicine.”
One of the main problems, he said, referring to the gap in data, is that there’s no specific information supplied about when the antibiotics were administered to the animals, how and why.
This would bring to light why antibiotics are being given to the animals in the first place. In 1973, the FDA ruled against using medically important antibiotics to promote growth in animals. However, they are allowed to be used to prevent disease as would be the case if some of the animals in a herd were ill or they had gone through stress through shipping or hot weather, for example.
Unfortunately, they’re often used to prevent diseases when there are no sick animals in the herd.
Since 2016, FDA officials have been saying that they wanted to get more concrete “duration limits” — how long certain antibiotics can be used — on the labels of antibiotics put into animal feed. This year, the agency put forward a draft “concept paper” for how duration limits might come about for different antibiotics already on the market.
However, Wallinga said that the ”flawed approach” depends too much on drug companies to set their own limits, and even then would allow supposed limits that instead would be too vague and meaningless to offer the needed level of public health protection.
Administering antibiotics for a longer time than needed, which is often the case with livestock, can trigger antibiotic resistance, said Wallinga. “The more we use them, the faster they’ll become ineffective. Long-term, it will be the superbugs rather than us that benefit,” he said.
This doesn’t translate well to the U.S. livestock industry, where many if not most medically important antibiotics are routinely put into animal feed for entire herds for so-called disease prevention, said Wallinga.
He said he believes that FDA’s approach has been primarily to get the livestock industry to voluntarily decrease antibiotic reliance, but he also points out that in the United States, three or four companies control 80 percent of production.
That’s why he believes “comparable and integrated analyses” covering both sales and use of medically important antibiotics are urgently needed.
Such analyses are essential, he says, for policymakers and the public to understand more precisely how these precious medicines are being used on farms and feedlots, and whether U.S. efforts to curb unnecessary use is succeeding or falling short.
Wallinga said that since 2009, the FDA has reported annual sales of livestock antibiotics but no information on veterinary prescriptions or orders. At the same time, human sales data also are not reported. Meanwhile, the CDC now issues annual reports on antibiotic prescriptions in human medicine, but these reports do not provide national data on medical antibiotic sales.
Providing the public each year with side-by-side comparisons of the use of these precious medicines in human medicine and in food-producing animals, is an important step, said Wallinga.
He said that these additional essential steps should also be followed:
- Ensuring that livestock producers are required to report annually on their on-farm use of antibiotics.
- Making critical investments to create a national system that collects, integrates, and publishes annual data on antibiotic use and antibiotic resistance, both in human and animal settings.
- Setting national targets for reducing medically important antibiotic use, especially in specific livestock sectors such as the beef and pork industries where most such antibiotics are now used.
Unlike Canada, the United Kingdom, Denmark or the Netherlands, the U.S. government does not release annual reports that integrate the human and animal sides of the antibiotic resistance problem. Wallinga said countries that do release such information have seen cases of resistance decline.
Some good news from the FDA
In recent days, the U.S. Food and Drug Administration’s Center for Veterinary Medicine published a report summarizing antibiotics sold or distributed for use in food-producing animals in 2020. It puts out a report on this topic each year.
It had some good news to share. It shows that domestic sales and distribution of medically important antimicrobial drugs approved for use in food-producing animals decreased by three percent between 2019 and 2020.
That represents a 38 percent decrease since 2015, which was the peak year of sales.
“This suggests that continued efforts to support the judicious use of antimicrobials in food-producing animals are having an impact,” says the report.
According to the agency, although sales data on antimicrobial drug products intended for food-producing animals do not necessarily reflect the actual use of antimicrobial drugs, sales volume observed over time can be a valuable indicator of market trends related to these products.
It also points out that sales and distribution information does not represent actual use of the products.
And it reminds the public that the agency’s goal in all of this is to to slow the development of antimicrobial resistance and preserve the effectiveness of antimicrobials for fighting disease in animals and humans.
“The agency’s aim is not simply measured by a reduction in sales volume of antimicrobials but also includes fostering good antimicrobial stewardship practices by optimizing the use of these products and limiting their use in animals to only when necessary to treat, control, or prevent disease.”
Good farming tactics
Wallinga said that every country in the European Union is already collecting information about antibiotic use on farms, or will soon be legally required to do so.
“In Europe, it was the federal officials telling livestock producers the direction they need be going,” he said, pointing out that the approach there is to promote healthier animals so as to prevent disease and therefore avoid antibiotics in the first place. In fact, next month the use of antibiotics for disease prevention absent any disease, will become illegal in livestock production in Europe. By contrast, he said, FDA not only allows it, but goes one step further by claiming that such use is “therapy.”
“By now, many of the largest livestock-producing countries in Europe have reduced their use of antibiotics by 50 to 60 percent in the last 10 years,” Wallinga said.
Here in the United States, many sustainable farmers rely on good farming practices instead of antibiotics, to keep their animals healthy.
“If we do that, why would we need antibiotics. unless, of course, an animal is actually sick,” said Linda Neunzig, a sheep farmer in Western Washington.
Organic producers, meanwhile, are not allowed to use antibiotics, or, if they do, in the case of treating a sick animal or bird, they can’t sell the meat, milk or dairy products as “organic.”

Dairy farmers who treat their cows with antibiotics can’t sell milk from those animals until the animals’ systems are clear of them.
For George Vojkovich, co-owner of Skagit River Ranch, which produces and sells pasture-raised organic beef, broilers, pork and eggs, it all comes down to good animal husbandry. His farming practices include building the soil, culling animals with genetic tendencies for certain health problems, using selective breeding, rotational grazing, planting a diversity of grasses and legumes and making sure the animals get the trace minerals they need.
Oregon organic dairy farmer John Bansen, said that raising healthy cows is a matter of making sure that the good bacteria outnumber the bad bacteria.
“It can’t be about ‘no bacteria,’ ” he said. He also believes that when the bacteria in the gut is healthy, the whole cow is healthy.
“You don’t want to subtract from a cow’s natural immune function,” he said. “Our job isn’t to replace her immune system with antibiotics and vaccines.”
Although many people believe that smaller “family-size” farms are the only places where animals and poultry can be raised without an excessive use of antibiotics, veterinarian Charles Hofacre, formerly a professor at the University of Georgia’s Center for Food Safety in the Avian Medicine Department, would disagree.
In an earlier interview with Food Safety News he said that raising animals and poultry in large-scale farming operations without pumping them full of antibiotics is being done every day and has been done for the 30 years that he’s been a poultry veterinarian.
And Wallinga is quick to praise Perdue Farms for being one of the first large companies to say that it will move away from antibiotic use, “No antibiotics ever” say the labels on its chickens and turkeys,
On a broader scale, the CDC offers this perspective about what it refers to as antibiotic stewardship:
“Perhaps the single most important action needed to greatly slow down the development and spread of antibiotic-resistant infections is to change the way antibiotics are used. Up to half of antibiotic use in humans and much of antibiotic use in animals is unnecessary and inappropriate and makes everyone less safe.
“Stopping even some of the inappropriate and unnecessary use of antibiotics in people and animals would help greatly in slowing down the spread of resistant bacteria,” according to the CDC. “This commitment to always use antibiotics appropriately and safely — only when they are needed to treat disease, and to choose the right antibiotics and to administer them in the right way in every case — is known as antibiotic stewardship.”
CDC’s advice to livestock and poultry producers:
- Only give antibiotics to animals under veterinary supervision.
- Don’t use antibiotics for growth promotion or to prevent diseases in healthy animals.
- Vaccinate animals to reduce the need for antibiotics.
- Support the implementation of effective interventions put in place to reduce the spread of AMR through the environment.
A global prospective
Early this month, the World Health Organization came out with its own guidelines for national governments and the global food animal industry. No waffling here. The guidelines not only call for a prohibition on the use of medically important antibiotics for disease prevention but also urge significant limits be put on using critically important antibiotics in food animals to treat diseases.
Here are the new guidelines:
- Reduce overall use of all medically important antimicrobials in food animals.
- Fully prohibit use of all medically important antimicrobials in food animals for growth promotion.
- Fully prohibit use of all medically important antimicrobials in food animals for preventing infectious diseases that have not yet been clinically diagnosed by a veterinarian.
- Prevent use of critically important antibiotics in food animals for disease control and use of highest priority critically important antibiotics in food animals for disease treatment.

“We have consistently advocated for FDA to better align domestic policies with the WHO and to take stronger measures to restrict and reduce use of antibiotics in food animals raised in the U.S.,” said Cameron Harsh, senior manager for Organic & Animal Policy for the Center for Food Safety. “The new guidelines illustrate the degree to which our regulators and large food animal producers are falling short.”
Harsh also said that the Center for Food Safety “will continue to advocate for best practices that include stocking densities that allow for full freedom of movement and expression, adequate access to sunlight and the outdoors, appropriate and nutritious diets, and breeding for traits that prioritize health and immunity benefits over growth and productivity.”
What about labels?
Food labels such as“No Antibiotics Ever (NAE)” or “Raised Without Antibiotics,” or “No Added Antibiotics” are used by farmers who voluntarily avoid using antibiotics on their livestock or poultry. When these labels accompany a seal that states “USDA Process Verified,” it means USDA inspectors visited the farm to verify antibiotic use on that farm.
Symptoms mimic foodborne illness
Symptoms of antibacterial infection are similar to symptoms foodborne illnesses but often more severe:
- Bloody diarrhea
- Fever over 102 degrees F, measured by mouth
- Frequent vomiting that prevents keeping liquids down
- Signs of dehydration, including little or no urination, a very dry mouth and throat, or feeling dizzy when standing up
- Diarrhea that lasts more than 3 days
In cases like this, medical attention should be sought.
Food safety advice for consumers
Go here https://urldefense.com/v3/__http://battlesuperbugs.com/article-archive/new-arac-video-safe-handling-meat-and-poultry-products__;!!NO21cQ!Th3N6fotqpDJCEV0mge-8QFJbUVj3tagnOcJjZt5CG38HlxwDggs2SPyIc4g4g$ for some advice from the Antibiotic Resistance Action Center about choosing meat and poultry in the store and preparing it in the kitchen. One of the points is to look for labels. Another is to avoid eating raw or undercooked meats.
An important priority
“Pandemic preparedness and public health protection should be our nation’s foremost priority,” said Wallinga. “We must therefore invest to robustly track antibiotic resistance and antibiotic use wherever it occurs.”
(To sign up for a free subscription to Food Safety News, click here.)









