Two federal programs are coordinating on $20 million in prizes for all phases of competition for new, innovative, and novel laboratory diagnostic tests to combat the development and spread of drug-resistant bacteria. Announced Sept. 8 by the National Institutes of Health and the HHS Office of the Assistant Secretary for Preparedness and Response, the competition — the Antimicrobial Resistance Diagnostic Challenge — is designed to tackle what officials call “a rising public health threat.”
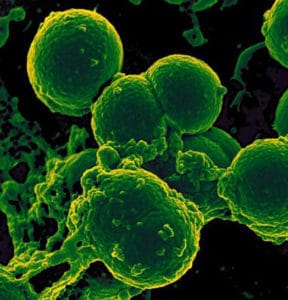
“The growing incidence of serious infections from antibiotic-resistant bacteria presents a critical risk to the public health of our nation,” said NIH Director Francis S. Collins, M.D., Ph.D. “My hope is that this competition will spur exceptional innovators to rise to the challenge and deliver effective tools to help manage this significant problem.” Antibiotic-resistant bacteria cause at least 2 million infections and 23,000 deaths each year in the United States, according to the Centers for Disease Control and Prevention (CDC). The problem was further underscored last week when CDC reportedly found a strain of E. coli with the antibiotic-resistance gene known as mcr-1 in a two-year-old Connecticut girl. She is the fourth human case identified in the U.S. so far. Diagnostic tests being sought in the federal awards competition are those that identify and characterize antibiotic-resistant bacteria and those that distinguish between viral and bacterial infections to reduce unnecessary uses of antibiotics, a major cause of drug resistance. With real-time detection, healthcare providers would be able to identify infecting pathogens and resistance factors within hours, rather than the two to three days or longer that the standard microbiological culture processes require, according to the award sponsors. Such knowledge would allow tailoring of treatments, minimizing the broad-spectrum antibiotic approach used by many clinicians today. The awards are being sponsored in support of the White House’s National Action Plan for Combating Antibiotic Resistant Bacteria. Concepts must be submitted by Jan. 9, 2017, for the first phase of the competition. Up to 20 semi-finalists will be selected from the applicant pool, each receiving up to $50,000. In the second phase of the competition, on Dec. 3, 2018, up to 10 finalists will be selected to each receive up to $100,000. These funds can be used to develop prototypes for evaluation by two CLIA-certified independent laboratories, which will be considered when final winners are selected. In the final phase, winners are expected to be announced on July 31, 2020. The competition specifies that up to three winners can be selected, and winners will share an amount equal to or greater than $18 million. For more information about the challenge or how to apply, go here.
(To sign up for a free subscription to Food Safety News, click here.)








