Nutricia infant formula has been found to contain Cronobacter after testing in Australia.
A batch of KetoCal 3:1 was positive for Cronobacter spp. during testing at the border by customs officials. There have been no illnesses linked to the product, and the source of contamination is being investigated by the company.
The implicated formula is a specialist product not sold to the general public but available under prescription for children with special nutritional needs.
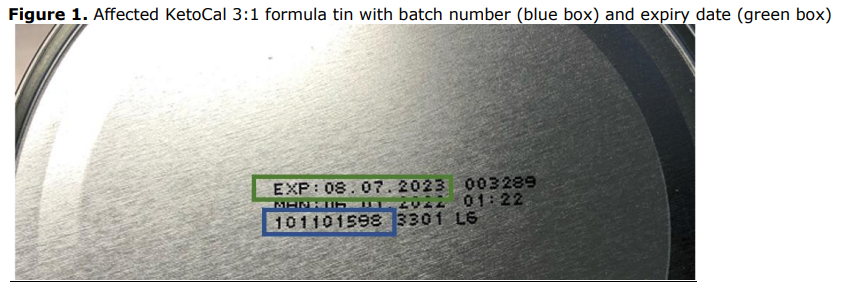
Affected tins are batch number 101101598 with best before July 8, 2023. For many patients there is no alternative feeding option so Nutricia has provided advice to consumers on how to make the product safe.
Revised preparation instructions
Nutricia, which is owned by Danone, recommends preparing the affected batch of KetoCal 3:1 by replacing cool water with water at a temperature of 80 degrees C (176 degrees F).
Fresh water should be boiled for 5 minutes and allowed to cool to 80 degrees C (176 degrees F) and measured with a sterilized thermometer. Nutricia has offered to send thermometers to people but boiled water should take about 4 minutes to cool down. Before feeding the baby, test the formula’s temperature by putting a few drops on the inside of your wrist. It should feel warm, not hot.
People were also told to wash hands with soap and water before preparing the infant formula and to clean surfaces such as countertops and sinks, with soap and water, or use a disinfectant wipe or paper towel sprayed with a cleaning product.
In a letter to healthcare professionals dated April 29, Nutricia said it was committed to providing customers with the highest quality medical nutrition items and that all products undergo rigorous testing before being released for sale.
Nutricia said there is currently only around 600 tins of the affected batch available in Australia and the next shipment of 800 tins from this batch was due around May 23. A new batch from Europe has been ordered with the expected arrival being May 7.
It is normal procedure for Australian customs to do random routine testing of imported products.
Public advice
Cronobacter infection in infants will usually start with a fever and poor feeding, excessive crying, very low energy or a grunting sound while breathing. Some may also have seizures. If your child develops these symptoms, take them to a doctor as soon as possible. Those more likely to get sick from Cronobacter infections include infants 2 months and younger, those born prematurely or with weakened immune systems, such as those undergoing chemotherapy.
People were advised not change or stop their child’s formula without consulting their specialist pediatrician, neurologist, metabolic physician or dietitian. Parents and carers who want an alternative nutrient source were told to speak to an expert to understand what options were able to be prescribed to meet the baby’s needs.
It is the second case of Cronobacter contamination in infant formula identified in Australia in 2022.
Various batches of EleCare, Similac and Alimentum infant formula made by Abbott Nutrition were recalled in February following illnesses and two deaths in infants who consumed the products in the United States. According to a statement from the company, Abbott tests products prior to distribution, and no Abbott formula distributed to consumers tested positive for Cronobacter sakazakii or Salmonella.
No cases related to the Abbott products have been recorded in Australia. The Abbott products were available internationally and in Australia through through prescriptions at pharmacies, hospitals and via direct mail order. The U.S. Food and Drug Administration continues its investigation into the Abbott situation.
(To sign up for a free subscription to Food Safety News, click here.)








