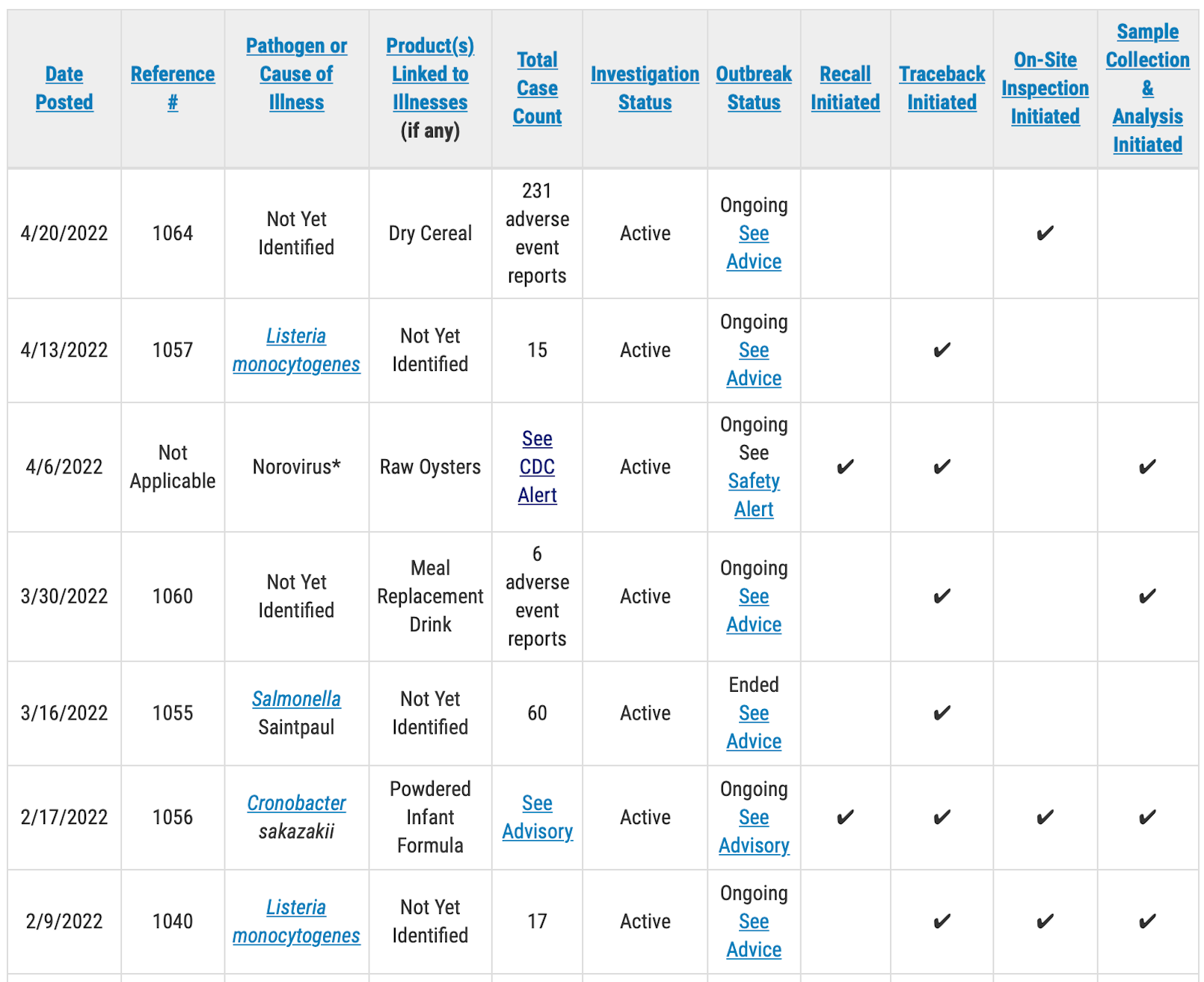
The FDA is investigating more than 200 “adverse events” related to an unnamed dry cereal. In recent days reports from across the country regarding illnesses linked to Lucky Charms cereal have been filed with government agencies and the iwaspoisoned.com website.
The reports include vomiting, diarrhea and other gastrointestinal symptoms. The Food and Drug Administration has separately reported in recent days that is is investigating complaints about Lucky Charms but has not released any other details about the investigation. General Mills, the maker of Lucky Charms, has reported that it is not aware of any confirmed illnesses associated with the cereal.
As of April 20, the FDA has initiated an on-site inspection in relation to the complaints about the cereal.
The agency has not reported any information about the people who made the complaints of adverse events related to the cereal and has not reported where they live.
In a statement released about the outbreak of “adverse events” linked to the “dry cereal,” the FDA states: “For adverse event report investigations, FDA will indicate a product category and not publicly name a specific product until there is sufficient evidence to implicate that product as a cause of illnesses or adverse events.
“For the new adverse events investigation — reference #1064 — FDA is following up on a series of unconfirmed adverse event reports (231 complaints) that may be associated with dry cereal. Although FDA has not determined that this cereal is linked to these adverse event reports, FDA is conducting an investigation to determine the potential causality of these complaints. The total number of adverse events reported includes the number of adverse events that have been self-reported by consumers to FDA consumer complaint coordinators and the CFSAN Adverse Event Reporting System (CAERS), which could include duplicate reports.”
For information about how to file a non-emergency food problem with the FDA, please click here.
In another investigation into different reports of “adverse events,” the FDA has revised the number of complaints to six, down from the 38 reported a week ago. The agency says the total number of complaints has been reduced to only reflect the events reported by consumers to the FDA’s complaint coordinators. The outbreak update did not indicate where the other 32 complaints had been filed.
The agency has identified the implicated product as a “Meal Replacement Drink,” but has not reported a brand or a distribution network. The FDA has begun traceback efforts on the product in question and has begun collection and testing of product samples.
Other ongoing outbreaks
Federal officials are investigating an outbreak of Listeria infections and are testing product samples in relation to another outbreak caused by Listeria monocytogenes.
The new Listeria outbreak has sickened at least 15 people, but the Food and Drug Administration has not yet identified a food source for the pathogen. In keeping with its usual process, the FDA has not released any information about the patients, such as age, and has not reported where they live.
As of April 20 the FDA had begun traceback but had not undertaken any on-site inspections or sample testing in relation to the outbreak.
In another outbreak, also caused by Listeria, the FDA is reporting that a food source has not been identified, but it has begun on-site inspection of an unnamed company. The agency has begun sample collection and testing, but it has not reported what is being tested. The patient count in the outbreak remains at 17.
The agency also has ongoing investigations into an outbreak of norovirus infections traced to raw oysters from British Columbia, Canada, and an outbreak of Salmonella Saintpaul infections from an unknown source. The norovirus outbreak traced to the oysters has sickened more than 100 people in the United States and more than 300 in Canada.
The FDA is also continuing to investigate an outbreak of cronobacter infections linked to infant formula made by Abbott Nutrition that has sickened four babies with two deaths under investigation. All production at the implicated production facility in Sturgis, MI, has been stopped.
The table below shows information about outbreak investigations being managed by FDA’s CORE Response Teams. The investigations are in a variety of stages. Some outbreaks have limited information with active investigations ongoing, others may be near completion. The table below has been abbreviated to show only active investigations.
A public health advisory will be issued for investigations that have resulted in specific, actionable steps for consumers to take to protect themselves, according to the FDA. Please direct your attention to those pages for the most up to date information on the investigation and for consumer protection information.
Outbreak and adverse event investigations that do not result in specific, actionable steps for consumers may or may not conclusively identify a source or reveal any contributing factors. Adverse event investigations rely on self-reported data. Although these reports may name a particular product, FDA will only indicate a product category in the table and will not publicly name a specific product until there is sufficient evidence to implicate that product as a cause of illnesses or adverse events. If a cause and/or contributing factors are identified that could inform future prevention, FDA commits to providing a summary of those findings.
To view the FDA page with links to specific information on individual outbreaks, please click here.
(To sign up for a free subscription to Food Safety News, click here)