Recalls are being issued worldwide for powdered infant formula, including Similac, following the announcement of recalls in the United States and Canada.
Those recalls include actions in Singapore and New Zealand and are related to an outbreak in the United States that has seen at least four children hospitalized. One death is being investigated by U.S. officials. Three of the infections are from cronobacter sakazakii and one is from Salmonella.
The implicated infant formula products are from an Abbott Nutrition facility in Sturgis, MI, according to the U.S. Food and Drug Administration.
The company has initiated a recall for its Similac, Alimentum and EleCare manufactured at the Sturgis, MI, production plant.
Recalled products were distributed to the following countries in addition to the United States: Australia, Bahrain, Barbados, Bermuda, Canada, Chile, China, Colombia, Costa Rica, Dominican Republic, Ecuador, Egypt, Guam, Guatemala, Hong Kong, India, Indonesia, Israel, Jordan, Kuwait, Lebanon, Malaysia, Mexico, New Zealand, Oman, Peru, Puerto Rico, Qatar, Saudi Arabia, Singapore, South Africa, Sudan, Taiwan, Thailand, United Arab Emirates, United Kingdom, and Vietnam ANI South.
The FDA’s findings to date include several positive cronobacter results from environmental samples taken at the Abbott facility in Sturgis, MI. A review of the Abbott ’s internal records also indicate environmental contamination with cronobacter sakazakii.
Abbott previously destroyed product because of the presence of Cronobacter sakazakii, according to the U.S. Food and Drug Administration.
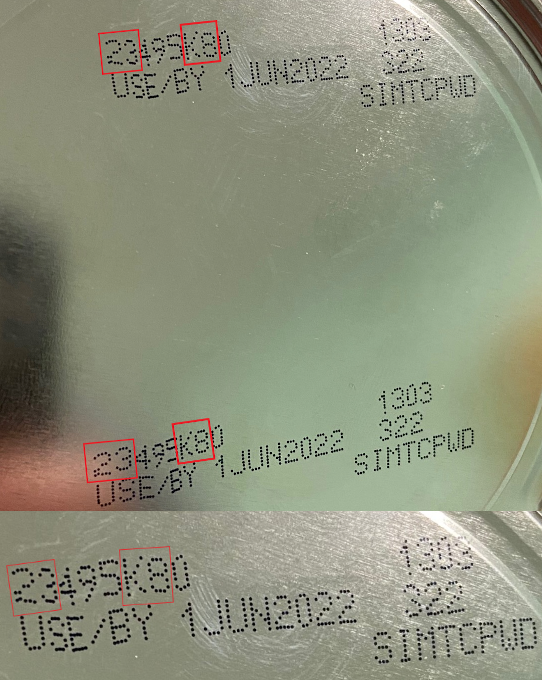
The FDA is advising consumers not to use Similac, Alimentum, or EleCare powdered infant formulas if:
- The first two digits of the code are 22 through 37; and
- The code on the container contains K8, SH or Z2; and
- The expiration date is 4-1-2022 (APR 2022) or later.
State and local health departments across the United States are warning consumers about the recalled infant formulas.

The Minnesota Department of Health investigated a case of an infant who was sickened by Cronobacter sakazakii in September 2021, according to Politico.
State health officials in Minnesota discovered that the infant had consumed powdered formula produced at the implicated Abbott facility and shared this information with FDA and CDC in September, the agency said. The Minnesota baby who got sick in September survived but was hospitalized for 22 days, state officials told Politico.
Cronobacter bacteria can cause severe, life-threatening infections (sepsis) or meningitis (an inflammation of the membranes that protect the brain and spine). Symptoms of sepsis and meningitis may include poor feeding, irritability, temperature changes, jaundice (yellow skin and whites of the eyes), grunting breaths and abnormal movements. Cronobacter infection may also cause bowel damage and may spread through the blood to other parts of the body.
Salmonella are a group of bacteria that can cause gastrointestinal illness and fever called salmonellosis. Most people with salmonellosis develop diarrhea, fever and abdominal cramps. More severe cases of salmonellosis may include a high fever, aches, headaches, lethargy, a rash, blood in the urine or stool, and in some cases, may become fatal.
(To sign up for a free subscription to Food Safety News, click here.)









