Another person has been added to the patient toll in an outbreak of Listeria infections of unknown origins, according to the Food and Drug Administration.
As of Feb. 17 there have been 14 people confirmed as patients in the Listeria outbreak that was first announced by the FDA on Feb. 9. The agency has not reported any information on the ages of the patients or where they live.
FDA investigators have begun traceback efforts in relation to the outbreak of Listeria infections, but they have not reported what food or foods they are looking at.
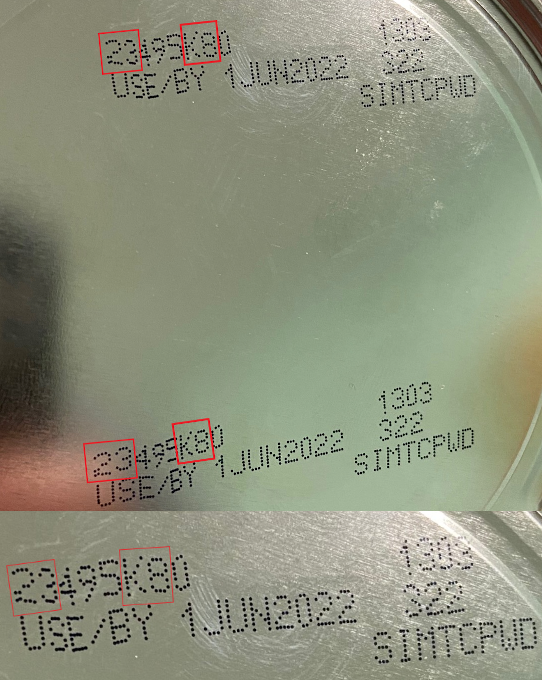
In a new outbreak listing the FDA reports that four infants have been infected from Cronobacter and Salmonella bacteria in infant formula products. All four patients were hospitalized.
One child with an infection from coronobacter has died. The death remains under investigation.
The FDA is advising consumers not to use Similac, Alimentum or EleCare powdered infant formulas if:
- the first two digits of the code are 22 through 37 and
- the code on the container contains K8, SH, or Z2, and
- the expiration date is 4-1-2022 (APR 2022) or later.
The company has initiated a recall.
All of the sick infants are reported to have consumed powdered infant formula (IF) produced from Abbott Nutrition’s Sturgis, MI, facility. The FDA has initiated an onsite inspection at the facility.
The table below shows information about four other ongoing outbreak investigations being managed by FDA’s CORE Response Teams and one investigation that has been closed. The investigations are in a variety of stages. Some outbreaks have limited information with active investigations ongoing, others may be near completion. The table below has been abbreviated to show only active investigations.
The Food and Drug Administration will issue public health advisories for outbreak investigations that result in “specific, actionable steps for consumers — such as throwing out or avoiding specific foods — to take to protect themselves,” according to the outbreak table page.
Not all recalls and alerts result in an outbreak of foodborne illness. Not all outbreaks result in recalls.
Outbreak investigations that do not result in specific, actionable steps for consumers may or may not conclusively identify a source or reveal any contributing factors, according to CORE’s outbreak table page. If a source(s) and/or contributing factors are identified that could inform future prevention, FDA commits to providing a summary of those findings, according to CORE officials.
(To sign up for a free subscription to Food Safety News, click here.)









