The Centers for Disease Control and Prevention have declared that the outbreak of infant botulism traced to ByHeart powdered formula is over.
Initially it was thought that there were 51 patients, but further review showed that three of the babies were sick from another source, bringing the total of outbreak cases to 48 spread across 17 states from coast to coast. All 48 infants were hospitalized but none died.
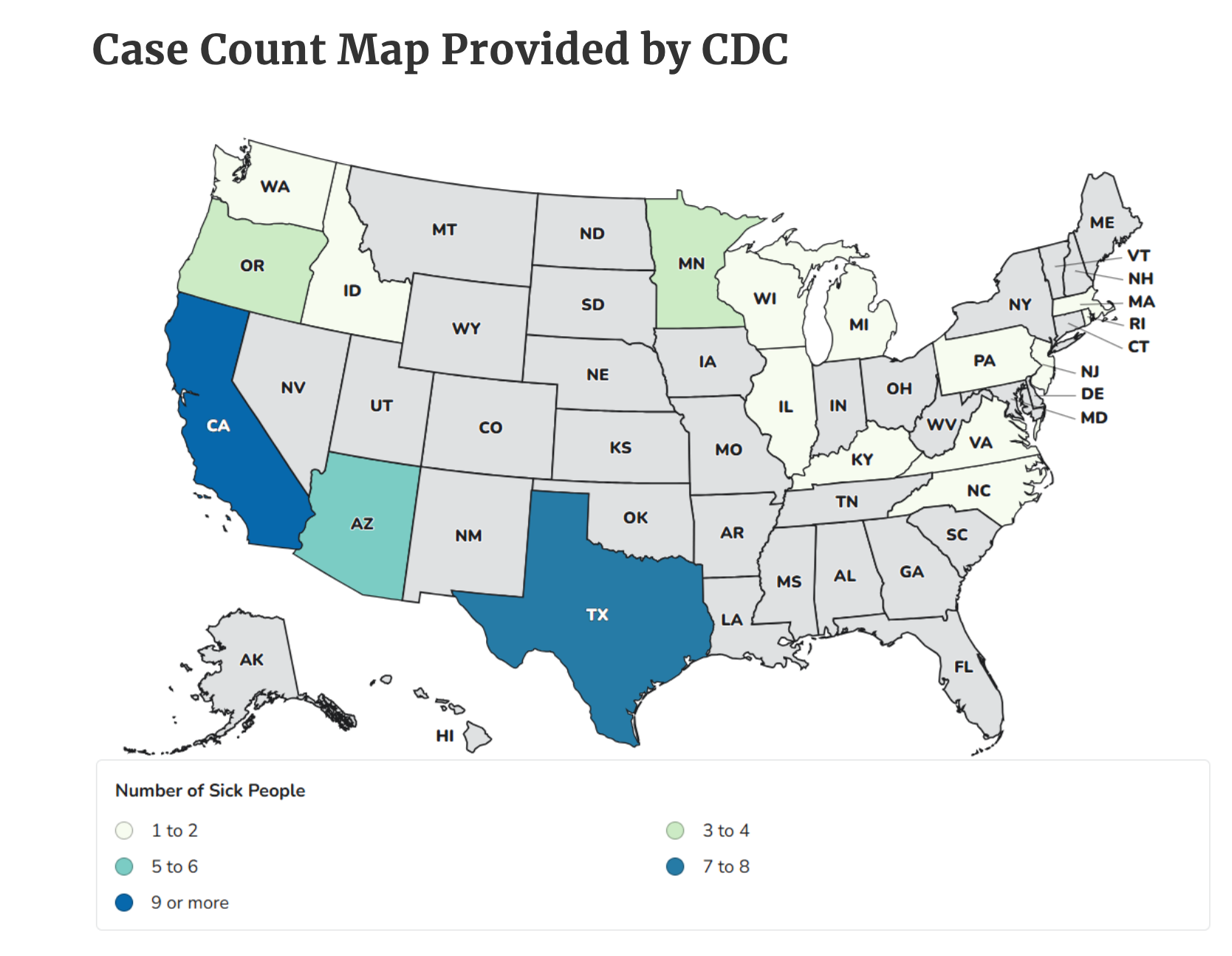
The vast majority of the babies became ill between August and November 2025, but investigators found that at least one patient had illness onset in March of 2022. The implicated company, ByHeart, began sales of its powdered infant formula in December 2023.
Lab testing detected botulinum neurotoxin type A and C botulinum type A in an opened container of the formula associated with one infant. Whole-genome sequencing showed genetic matches between bacteria isolated from infant stool samples and ByHeart powdered formula products, strengthening evidence of a common source.
The investigators also identified an atypical geographic pattern of toxin types, including rare type A cases in parts of the country where type B disease is usually dominant — another signal that prompted federal and state agencies to treat the situation as a multistate outbreak, according to the Center for Infectious Disease Research and Policy.
The California Department of Public Health (CDPH), Infant Botulism Treatment and Prevention Program (IBTPP) was the first entity to identify the outbreak. The Food and Drug Administration assisted the CDC and California officials with the outbreak investigation.
The FDA found that two isolates from one lot of organic whole milk powder used to produce the ByHeart infant formula matched two isolates from one lot of organic whole milk powder, collected and analyzed by FDA, have been shown by whole genome sequencing (WGS) analysis to match a cluster of isolates reported by FDA on Jan. 23, 2026. These two new isolates were collected by FDA at Dairy Farmers of America, the processor for Organic West Milk, which supplies ByHeart. This cluster now also includes:
- one closed powdered infant formula sample tested by ByHeart,
- one closed powdered infant formula sample tested by the New York State Department of Health, Wadsworth Laboratory,
- one clinical isolate,
- three isolates from one lot of organic whole milk powder tested by ByHeart.
Additionally, WGS analysis of clinical isolates has shown that two recently analyzed clinical isolates collected and analyzed by CDPH match clusters of isolates from product samples.
One clinical isolate matches a cluster that now includes:
- three clinical isolates and
- an open powdered infant formula sample (tested by CDPH and previously reported as confirmed positive on Nov. 18, 2025)
The second recently analyzed clinical isolate matches a cluster that now includes:
- one clinical isolate,
- four isolates from one lot of powdered infant formula tested by ByHeart and previously reported in FDA’s Jan. 23, 2026 update. This lot was reported by one patient, and
- one sample of base mix used to make powdered infant formula tested by ByHeart
The detection of Clostridium botulinum in infant formula, or ingredients, is complex, according to the FDA. To date, WGS analysis has identified 17 different strains of this bacterium in samples from patients, finished products and ingredients.
“While these sample results add to the available evidence needed to investigate the root cause of this outbreak, due to the complexities of Clostridium botulinum and limited scientific evidence currently available, FDA has not yet determined a root cause(s). Additional sample analysis and research is being conducted and is necessary to inform possible conclusions about this outbreak,” according to the FDA.







