— OPINION —
A few weeks ago I posted, “Putting the F – Food – back into the FDA.” I was wrong.
It is time to “GET THE F OUT OF THE FDA.” The F is Food. The D is Drugs. We need to separate the FDA functions between Food (food safety and human nutrition) and Drugs (and medical devices).

Last fall the credibility of the FDA was again on the butcher block. This time it was the FDA’s failure to adequately inspect an infant formula manufacturer which led to likely ill children, a recall, a plant closure and shelves bare of infant formula. Faced with angry parents and politicians, the leadership at the FDA opened an old playbook to avoid accountability – ask a panel to give recommendations.
Enter the Reagan-Udall Foundation.

The Reagan-Udall panel was made up of esteemed supporters of the FDA and its function. Those asked to testify before the panel (including myself) all had a deep desire to create a functioning FDA that would do its function at the highest levels. Click here for the full report.
My vision of a more empowered food side of the FDA would have created two Senate-appointed commissioners – one with a portfolio of all aspects of food as mentioned above and one with a portfolio of drugs and medical devices.
The first option of the Reagan-Udall panel is to “Create Separate Food and Drug Administrations within HHS.”
Pulling from the panel’s recommendations, the FDA’s path to a better food supply seems clear, albeit a lot to “chew on.”
Here is a chart from the report:
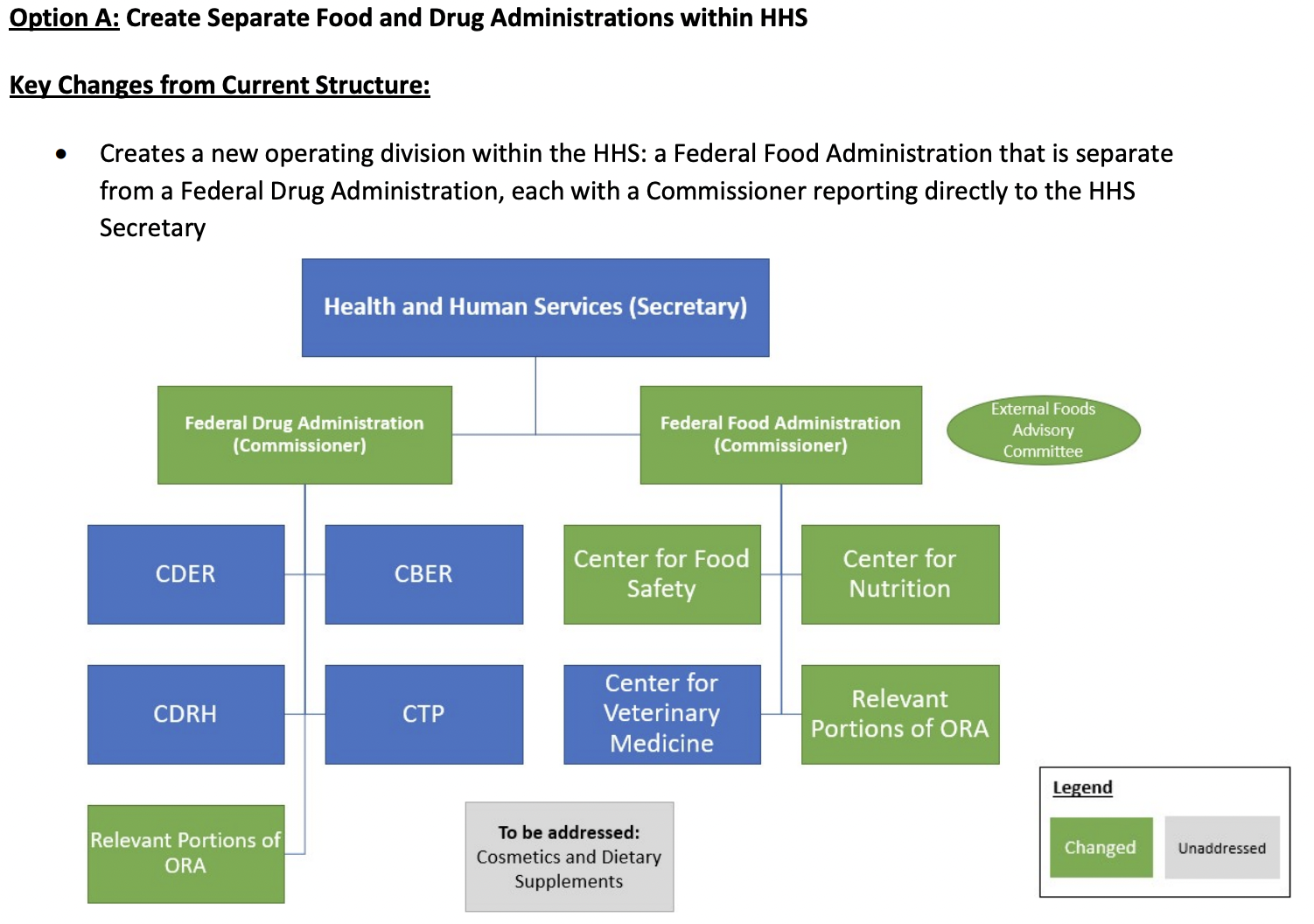
I believe the Human Foods Program should have clear lines of authority and accountability.
Within the Human Foods Program, the importance of nutrition should be elevated – although I might add, not at the expense of food safety.
The foods portfolio of the Office of Regulatory Affairs (ORA) – the inspection arm – should be integrated directly with the other elements of FDA’s Human Foods Program.
The food-relevant work of the Center for Veterinary Medicine (CVM) should be integrated with the overall FDA Human Foods Program.
A new Foods Advisory Committee, at the Commissioner level, should be established to strengthen external input to Human Foods Program activities.
Structure changes should be implemented with cultural transformation efforts.
Given the economic impact that foodborne illness and diet-related chronic disease have on Americans and the federal budget, it is imperative that the Human Foods Program become more prominent. When compared to the medical products programs within FDA, the Human Foods Program continuously struggles for visibility and prominence. A component of this elevation of the Human Foods Program is strong advocacy to advance the Human Foods Program at all levels of the government, especially at the Department of Health and Human Services (HHS) and the White House, including the Office of Management and Budget.
It is time the White House and Congress take responsibility for protecting the public by breaking the FDA apart and creating a new dedicated foods agency.
Go to www.marlerblog.com to check out my new “GET THE F OUT OF THE FDA” ad campaign.

(To sign up for a free subscription to Food Safety News, click here.)









