The FDA has closed investigations on two outbreaks and is continuing to investigate another eight.
An investigation into an outbreak of “adverse events” involving 558 patients who ate Lucky Charms cereal that was initially posted on April 20 is closed. The FDA conducted on-site inspections and testing.
“The investigation did not identify a pathogen or cause of the self-reported illnesses, despite extensive testing for numerous potential microbial and chemical adulterants,” according to the FDA.
An investigation into a Salmonella Braenderup outbreak initially posted on June 22 has been closed. A product linked to 75 reported illnesses was not identified
Meanwhile, patient counts in several other outbreaks have increased in the past week.
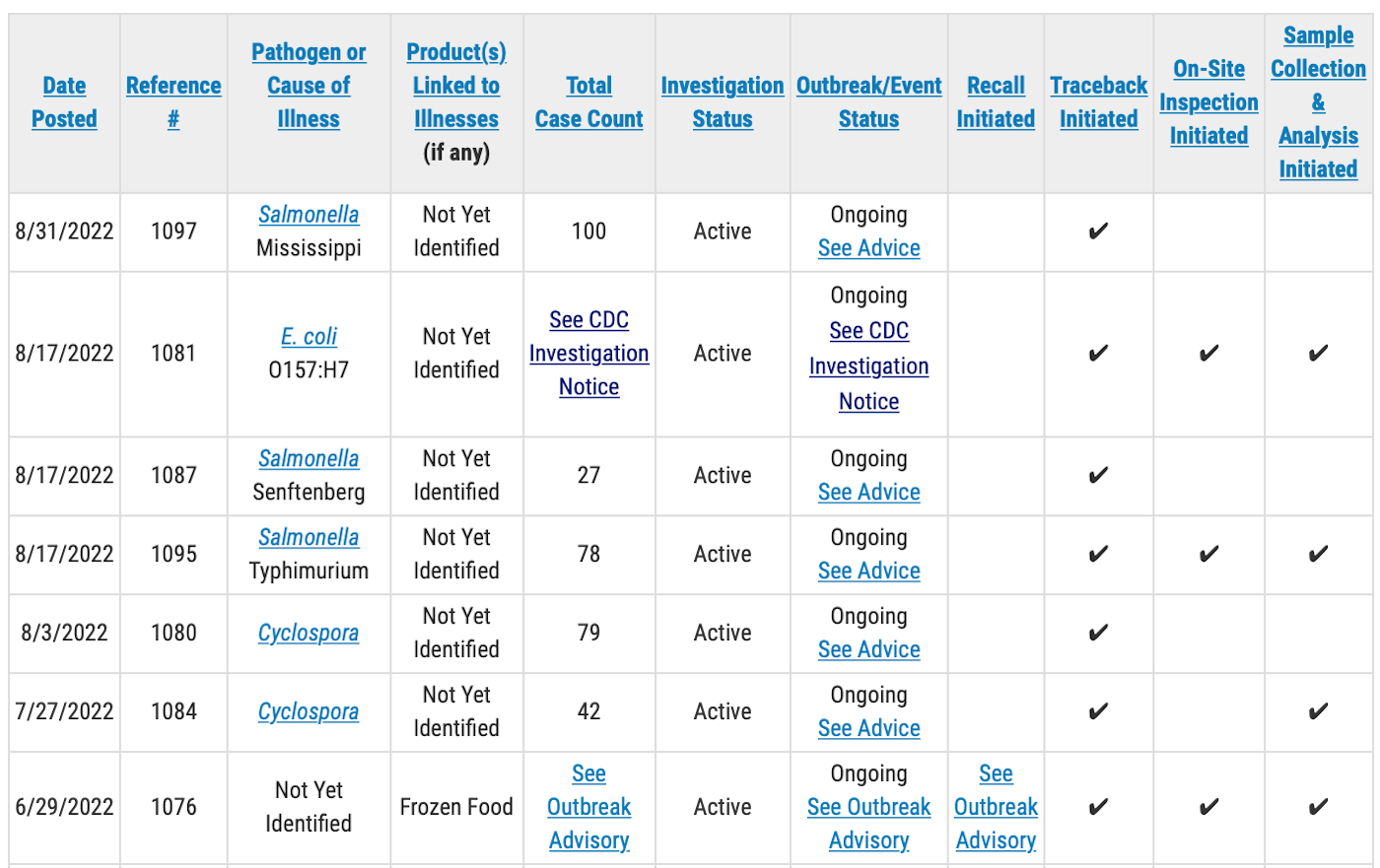
- For a Salmonella Mississippi outbreak from a not yet identified food, the patient count has increased from 99 to 100. Traceback has been initiated but the FDA has not reported what is being traced
- For a Salmonella Senftenberg outbreak from a not yet identified food, the case count has increased from 22 to 27 cases.
- For a Salmonella Mississippi outbreak from a not yet identified product, the case count has increased from 99 to 100 cases.
- For a Salmonella Typhimurium outbreak from a not yet identified food, the case count has increased from 73 to 78 cases. An on-site inspection and sample collection and analysis has been initiated, but the FDA has not reported what is being tested.
- For a Cyclospora outbreak the case count has increased from 75 to 79. Traceback has begun but the FDA has not reported what is being traced.
- For another Cyclospora outbreak the case count has remained steady at 42. Traceback has begun but the FDA has not reported what is being traced.
Other ongoing investigations
- An investigation related to adverse effects associated with Daily Harvest brand frozen Leeks & Lentils Crumbles is ongoing. The company has received more than 470 complaints of illnesses and as of its last report on July 29 the FDA had received 329 complaints. Some of the patients have gone into liver failure and at least 25 have had to have their gallbladders removed. The FDA is working on traceback efforts and has begun on-site inspection and product testing. Some testing has revealed that tara flour is an ingredient unique to the Daily Harvest crumbles product and could be related to the illnesses.
- An outbreak of infections from Cronobacter in four infants, one of whom died. The outbreak has been determined to be over by the CDC but is it still under investigation. The babies consumed infant formula made by Abbott Nutrition’s plant in Sturgis, MI.
Click here to go to the FDA page with links to specific outbreak details. The investigations are in a variety of stages. Some outbreaks have limited information with active investigations ongoing, others may be near completion.
A public health advisory will be issued for investigations that have resulted in specific, actionable steps for consumers to take to protect themselves, according to the FDA. Please direct your attention to those pages for the most up-to-date information on the investigation and for consumer protection information.
Outbreak and adverse event investigations that do not result in specific, actionable steps for consumers may or may not conclusively identify a source or reveal any contributing factors. Adverse event investigations rely on self-reported data. Although these reports may name a particular product, FDA will only indicate a product category in the table and will not publicly name a specific product until there is sufficient evidence to implicate that product as a cause of illnesses or adverse events. If a cause and/or contributing factors are identified that could inform future prevention, FDA commits to providing a summary of those findings.
(To sign up for a free subscription to Food Safety News, click here)









