How can Parents help prevent Cronobacter Infections now and in the future?
Do not use the powdered infant formula that is the subject of the recall.
The CDC has warned that powdered formula is not sterile. Prepare and store powdered infant formula safely. Make sure that your formula is not expired or recalled, and that the container is in good condition. Keep powdered formula lids and scoops clean, and close containers of formula as soon as possible. Prepare formula with hot water (at least 158°F/70°C) to protect against Cronobacter.
Breastfeed. Very few cases of Cronobacter infections have been reported among infants fed only breast milk. However, you need to clean, sanitize, and store feeding items and breast pump parts and baby bottles safely.
Consider using liquid formula when possible. If your baby gets formula, consider using formula sold as a liquid rather than a powder. This is especially important when your baby is less than 3 months old or if your baby was born prematurely or has a weakened immune system. Liquid infant formula is made to be sterile (without germs) and should not transmit Cronobacter infection when handled carefully.
Keep hands clean. Always wash your hands carefully with soap and water during key times:
- Before preparing and feeding bottles or foods to your baby.
- Before touching your baby’s mouth.
- Before touching pacifiers or other things that go into your baby’s mouth.
- After using the toilet or changing diapers.
What is Cronobacter?
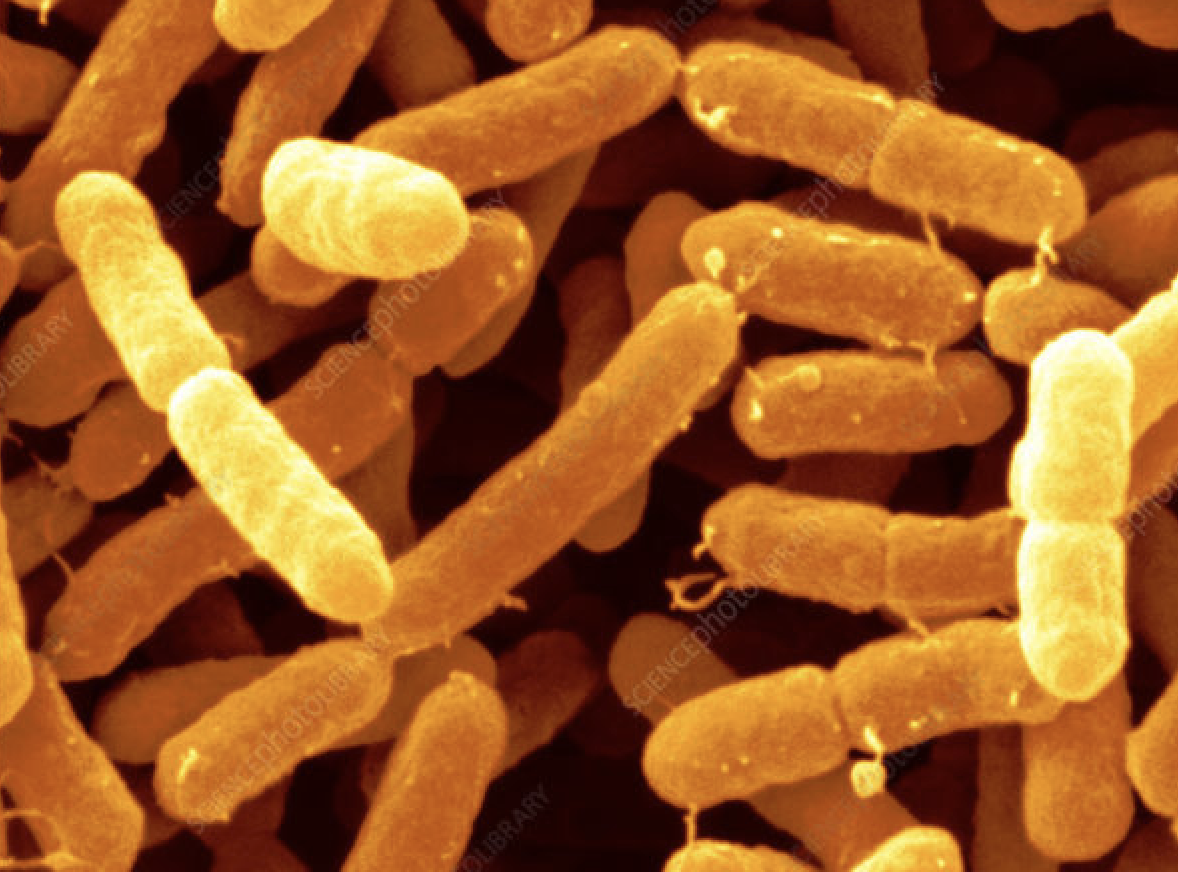
The CDC reports that Cronobacter infections are rare, but they can be deadly in newborns. Infections in infants usually occur in the first days or weeks of life. About two to four cases are reported to CDC every year, but this figure may not reflect the true number of illnesses because most hospitals and laboratories are not required to report Cronobacter infections to health departments.[1]
Cronobacter germs can cause a dangerous blood infection (sepsis) or make the linings surrounding the brain and spinal cord swell (meningitis). Infants 2 months of age and younger are most likely to develop meningitis if they get sick from Cronobacter. Other infants more likely to get sick are those born prematurely and those less able to fight germs and sickness because of illness or medical treatment, such as infants receiving chemotherapy for cancer.
The first symptom of Cronobacter infection in infants is usually a fever, accompanied by poor feeding, crying, or very low energy. You should take an infant with these symptoms to the doctor.
The Outbreak to Date.
The CDC and FDA report that from September 16, 2021, to January 5, 2022, CDC received reports of three Cronobacter cases in infants that were later found to be linked to FDA’s ongoing investigation. Since then, CDC has identified one additional case of Cronobacter infection in an infant who consumed formula produced at this facility.
- Four infants with Cronobacter infections in Minnesota (1), Ohio (2), and Texas (1) consumed formula produced at the Sturgis, Michigan, facility before they got sick.
- Cronobacter infections may have contributed to the deaths of two infants in Ohio.
Earlier, the FDA and CDC had identified a Salmonella Newport illness in an infant likely linked to the same infant formula. That link has been withdrawn.
What has been Recalled.

According to the FDA. on February 28, 2022, Abbott Nutrition recalled Similac PM 60/40 powdered formula (Lot # 27032K80 (can) / Lot # 27032K800 (case). The Similac PM 60/40 recall is in addition to other lots of Similac, Alimentum, and EleCare powdered formula that were recalled on February 17, 2022, for possible Cronobacter contamination.
Similac, Alimentum, or EleCare powdered formula products recalled on February 17 have all three of these notations:
- First two digits are 22 through 37, AND
- Code on the container contains “K8,” “SH,” or “Z2,” AND
- Use-by date is 4-1-2022 (APR 2022) or later
The recall impacts Alimentum, EleCare, and Human Milk Fortifier for markets outside the U.S. No other Abbott Nutrition products distributed outside of the U.S. are affected by this recall. According to the firm, recalled products were distributed to the following countries/locations: Australia, Bahrain, Barbados, Bermuda, Canada, Chile, China, Colombia, Costa Rica, Dominican Republic, Ecuador, Egypt, Guam, Guatemala, Hong Kong, India, Indonesia, Israel, Jordan, Kuwait, Lebanon, Malaysia, Mexico, New Zealand, Oman, Peru, Puerto Rico, Qatar, Saudi Arabia, Singapore, South Africa, Sudan, Taiwan, Thailand, United Arab Emirates, United Kingdom, and Vietnam ANI South.
Examples of Medical and Specialty Products
__________________
[1] Minnesota is the only state that requires Cronobacter cases to be reported. This lack of nationwide reporting may be a cause of the seemingly slow response by public health in this tragic outbreak.






